Emerging Trends in RegTech for Pharma SaaS

Key Takeaways
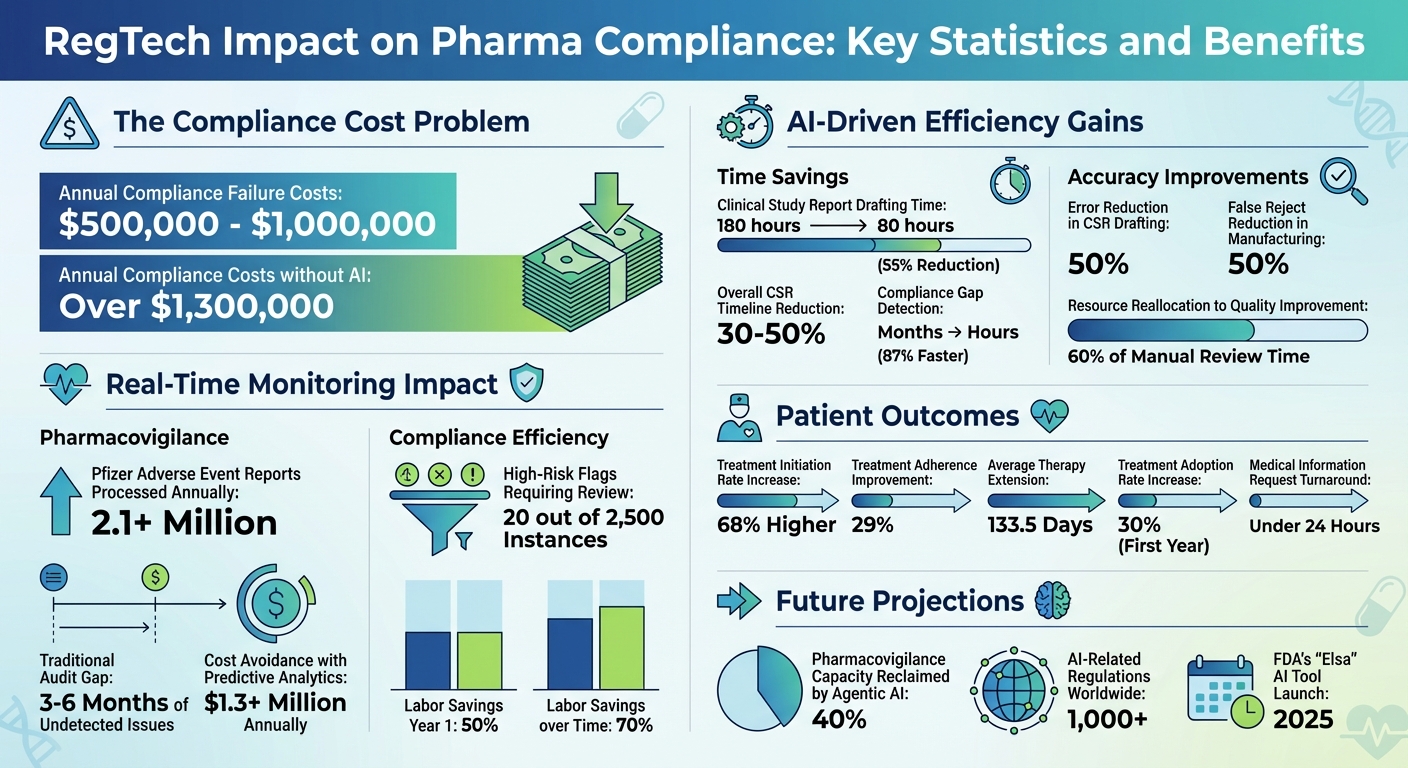
Pharma companies are under immense regulatory pressure, with compliance failures costing teams between $500,000 and $1 million annually. Enter RegTech: a solution leveraging AI and SaaS to automate compliance, reduce risks, and improve efficiency.
Key Takeaways:
- AI tools like NLP and Generative AI speed up compliance tasks, cutting Clinical Study Report timelines by up to 50%.
- Real-time monitoring replaces periodic audits, preventing costly delays in identifying issues.
- Predictive analytics reduce emergency consulting fees and help avoid compliance gaps.
- Patient-focused platforms integrate RegTech to boost treatment adherence by up to 30%.
RegTech is transforming compliance from reactive to proactive, saving time, money, and resources while improving patient outcomes.
RegTech Impact on Pharma Compliance: Key Statistics and Benefits
Beyond Compliance – Regulatory 4.0 and AI Strategies
sbb-itb-8f61039
AI-Driven Compliance Automation
Artificial intelligence is reshaping how pharmaceutical companies handle compliance. Gone are the days of solely relying on manual document reviews and periodic audits. AI now adds an analytical intelligence layer to existing Quality Management Systems, working in real time to improve efficiency without requiring a complete system overhaul. Below, we'll dive into the tools driving this shift and the measurable gains they deliver.
AI-Powered Compliance Tools
AI technologies like Natural Language Processing (NLP) for document analysis, Generative AI for drafting regulatory documents, and Autonomous AI for managing complex workflows are at the forefront of this transformation. These tools address regulatory challenges that manual processes often fall short on. For example, AI can differentiate between superficial deviation investigations and those with genuine scientific rationale and root cause analysis - an area of concern under 21 CFR 211.192.
One standout example is Pfizer's AI system, which categorizes and prioritizes Individual Case Safety Reports, allowing safety scientists to focus on more intricate analyses. Similarly, a collaboration between McKinsey and Merck showcased an AI platform that reduced the time to draft Clinical Study Reports from 180 hours to just 80 hours, while also cutting errors by 50%.
"Regulatory writing has outpaced what manual processes can handle. Automation is essential to keep up, so experts can focus on higher-value work." - Eunice Youhanna, Industry Advisor, Health & Life Sciences, Microsoft
These tools not only streamline processes but also improve regulatory outcomes, as highlighted in the next section.
Benefits of AI in Regulatory Processes
AI tools bring speed and cost-efficiency to compliance efforts, turning slow, manual reviews into streamlined operations. For instance, organizations using AI-powered compliance gap analysis report 87% faster gap detection, cutting issue identification from months to just hours. AI-assisted authoring has also slashed Clinical Study Report timelines by 30–50%. Financially, these tools help avoid annual compliance costs exceeding $1.3 million by reducing emergency consulting fees and mitigating warning letter risks.
AI doesn’t just save time - it reallocates resources effectively. Companies have redirected 60% of the time previously spent on manual reviews toward quality improvement initiatives. In manufacturing, Merck's use of generative AI for visual inspections between 2024 and 2025 led to a 50% reduction in false rejects, reducing waste and improving supply chain reliability. Beyond speed, AI uncovers patterns and inconsistencies across systems that manual reviews often miss, such as linking equipment maintenance issues to production deviations.
Real-Time Monitoring and Predictive Analytics
AI doesn’t just automate compliance - it changes the game entirely with continuous monitoring that prevents problems before they snowball. Traditional audits, often spaced months apart, leave a dangerous 3–6 month gap where flawed SOPs or batch records can slip through unnoticed. Real-time monitoring closes that gap, moving compliance from a reactive “fix-it-later” approach to proactive issue prevention. By combining AI-driven automation with constant oversight, companies can catch and resolve problems as they happen.
Real-Time Data Monitoring for Compliance
Pharma SaaS platforms are now leveraging real-time tracking to keep a pulse on production parameters, predicting quality outcomes before failures even occur. AI-powered machine vision systems, for example, inspect tablets and vials for defects like cracks, discoloration, or contamination with far greater accuracy than manual inspections. These systems have already demonstrated measurable efficiency gains, reducing waste and ensuring smoother supply chain operations.
This shift to continuous monitoring allows quality teams to focus their efforts where it matters most. Instead of combing through every batch record manually, review-by-exception systems flag deviations from validated parameters automatically. A great example is Pfizer, which has been using an AI system since 2014 to handle the overwhelming volume of adverse event case reports - now exceeding 2.1 million annually. The system categorizes reports and extracts the critical patient and drug data needed for compliance.
Predictive Analytics for Risk Management
AI doesn’t just stop at identifying current issues - it’s also looking ahead. Predictive analytics tools analyze historical data, such as FDA Form 483 observations and warning letters, to calculate the likelihood of compliance gaps leading to future inspection issues. With these tools, organizations report 87% faster detection of compliance gaps, turning what used to take months into a matter of hours. This speed saves companies big money, with cost avoidance exceeding $1.3 million annually by cutting down on emergency consulting fees and large-scale remediation efforts.
One of the most powerful aspects of AI is its ability to connect the dots across multiple systems. For example, it can link data from QMS, LIMS, EBR, and equipment maintenance software to uncover hidden patterns, such as how equipment maintenance problems might lead to production deviations. GlaxoSmithKline (GSK) has already seen success with an AI-driven compliance system that analyzes manufacturing and audit data. This system reduced documentation errors during internal audits and improved readiness for inspections.
There’s also a growing focus on algorithmovigilance, which involves monitoring AI systems themselves to ensure they stay accurate over time. This is especially critical for clinical decision-support tools, which must adapt as patient demographics and conditions evolve.
"AI compliance gap analysis is not a replacement for your Quality Management System (QMS). It's an intelligence layer that reads from your existing systems... and identifies what your QMS was never designed to detect." - Harsh Shah, Clinplex AI
Patient-Centric RegTech and SaaS Integration
RegTech, already known for its AI-driven compliance and predictive analytics, is expanding its role to improve patient outcomes. By combining these capabilities with patient-centric SaaS platforms, RegTech ensures that every step of the patient journey - from initial education to ongoing treatment adherence - meets strict regulatory standards while maintaining a personal, human connection.
Supporting Patient Adherence Through RegTech
RegTech introduces a Compliance Intelligence layer that keeps patient engagement workflows aligned with FDA 21 CFR and ICH guidelines. This ensures that tools supporting patients meet rigorous regulatory requirements. For instance, AI-powered systems can monitor patient–provider communications in real time, offering personalized support while staying compliant.
The results are impressive. Platforms like PatientPartner, which blend peer-to-peer mentorship with automated compliance, have achieved a 68% higher treatment initiation rate. PatientPartner pairs patients with experienced mentors who guide them through their healthcare journey, all while adhering to HIPAA and GDPR standards. Automated oversight ensures every interaction remains secure and compliant, which has led to a 29% improvement in treatment adherence and an average therapy extension of 133.5 days.
Digital tools also speed up response times. Chatbot-based policy apps allow representatives to deliver immediate, compliant answers to patient inquiries by referencing precise policy details in real time. This eliminates delays that might otherwise cause patients to abandon their treatment plans. Companies are working to reduce Medical Information Request Form turnaround times to under 24 hours, minimizing the risk of losing patient engagement opportunities.
These advancements highlight how RegTech can create scalable, compliant systems that improve adherence and patient support.
Scalable SaaS Features for Patient Engagement
For patient-centric platforms to succeed, they must scale effectively while maintaining compliance. Modern SaaS solutions achieve this by embedding RegTech capabilities into their core design rather than adding them later. Tools powered by real-time analytics and sentiment analysis use Natural Language Processing (NLP) to extract critical insights from patient interactions, flagging potential risks and enhancing engagement strategies.
This proactive approach to compliance monitoring has revolutionized patient program management. Instead of manually reviewing thousands of communications, NLP tools reduce the workload to just 20 high-risk flags out of 2,500 instances. This allows compliance teams to focus their expertise on critical areas while automation handles routine tasks. Companies adopting these systems report labor savings of 50% in the first year, increasing to 70% over time.
Security remains a top priority for these platforms. Adhering to standards like SOC 2, ISO 27001, and end-to-end encryption ensures patient data is secure while enabling the real-time monitoring RegTech demands. PatientPartner exemplifies this balance, combining robust security frameworks with features like AI-driven mentorship and data-powered sentiment analytics. Pharmaceutical companies trust such platforms to support patients at scale.
"Patient Partner is a unicorn in the industry, setting a new standard with their clear dedication to the mission of positively impacting patients' lives through innovation and compassionate support." - Melissa B., Sobi Pharmaceuticals
By integrating RegTech into patient-focused SaaS platforms, companies aren't just avoiding regulatory pitfalls - they're building trust. When patients feel confident their data is safe and their care is monitored for quality, they’re more likely to stay engaged with their treatment. Specialized platforms with built-in compliance have even driven a 30% increase in treatment adoption rates within the first year of implementation.
This seamless integration of compliance and patient engagement underscores RegTech's growing influence as both a regulatory tool and a catalyst for better patient outcomes.
Key Takeaways and Future Outlook
RegTech is reshaping pharma SaaS by shifting from reactive compliance to proactive intelligence. For instance, AI-powered automation can reduce the time needed to draft clinical study reports by an impressive 40% to 60%. Real-time monitoring is also revolutionizing pharmacovigilance, moving it from addressing past incidents to predicting risks before they occur. A notable development is the FDA's planned 2025 launch of "Elsa", a generative AI tool based on Anthropic's Claude model. This tool highlights how even regulators are adopting AI to speed up reviews and summarize adverse events. Together, these advancements are paving the way for faster and more precise drug development cycles.
In addition to improving processes, patient-focused platforms are leveraging these efficiencies to boost treatment adherence. Embedding RegTech capabilities directly into SaaS solutions allows companies to achieve tangible results, such as handling medical information requests in under 24 hours.
"Our minds are faster than our work allows us to be. We couldn't do certain things because the capability wasn't there. Now we can." - Sales Force Planning Executive, Large Pharma Company
This quote illustrates the growing shift toward fully integrated AI workflows, where technology enables what was previously impossible.
Looking ahead, advancements like agentic AI and algorithmovigilance promise to extend these benefits even further. Agentic AI, for example, is projected to reclaim 40% of pharmacovigilance capacity by automating complex workflows. Meanwhile, algorithmovigilance - continuous monitoring of AI systems to catch performance issues or data drift - will play a key role in maintaining reliability. With over 1,000 AI-related regulations introduced worldwide, companies that adopt "overlay" compliance intelligence platforms can stay agile without disrupting their existing quality management systems.
The future of RegTech in pharma SaaS is all about building trust, accelerating innovation, and improving patient outcomes. Organizations that embrace these technologies today will be better equipped to navigate an increasingly intricate regulatory environment.
FAQs
How do RegTech AI tools fit into my existing QMS without replacing it?
RegTech AI tools work seamlessly with your current Quality Management System (QMS), improving compliance processes without the need for a complete overhaul. They offer features like real-time monitoring, predictive analytics, and automated compliance gap analysis, which help simplify workflows and cut down on manual tasks. Acting as an additional layer, these tools boost efficiency and support better decision-making while ensuring compliance with regulatory standards such as GxP and FDA requirements - all without interfering with your existing QMS.
What data is required to start real-time compliance monitoring and predictive analytics?
To kick off real-time compliance monitoring and predictive analytics, you'll need a few key inputs: up-to-date regulatory updates, historical compliance records, document management details, and global regulatory insights. These elements work together to power AI-driven analysis, helping to simplify compliance processes and predict potential risks before they become issues.
How can PatientPartner add RegTech controls without slowing patient engagement?
PatientPartner uses AI-powered compliance tools and real-time monitoring to strengthen its regulatory controls. These features provide constant oversight, ensuring that patient engagement remains efficient and uninterrupted, all while delivering a smooth and hassle-free experience for patients.




