Cross-Functional Teams: Best Practices 2025

Key Takeaways
Cross-functional teams are transforming the pharmaceutical and med-tech industries. Instead of isolated departments, these teams combine diverse expertise to solve complex challenges like integrating AI, improving patient outcomes, and navigating regulations. Here’s what you need to know:
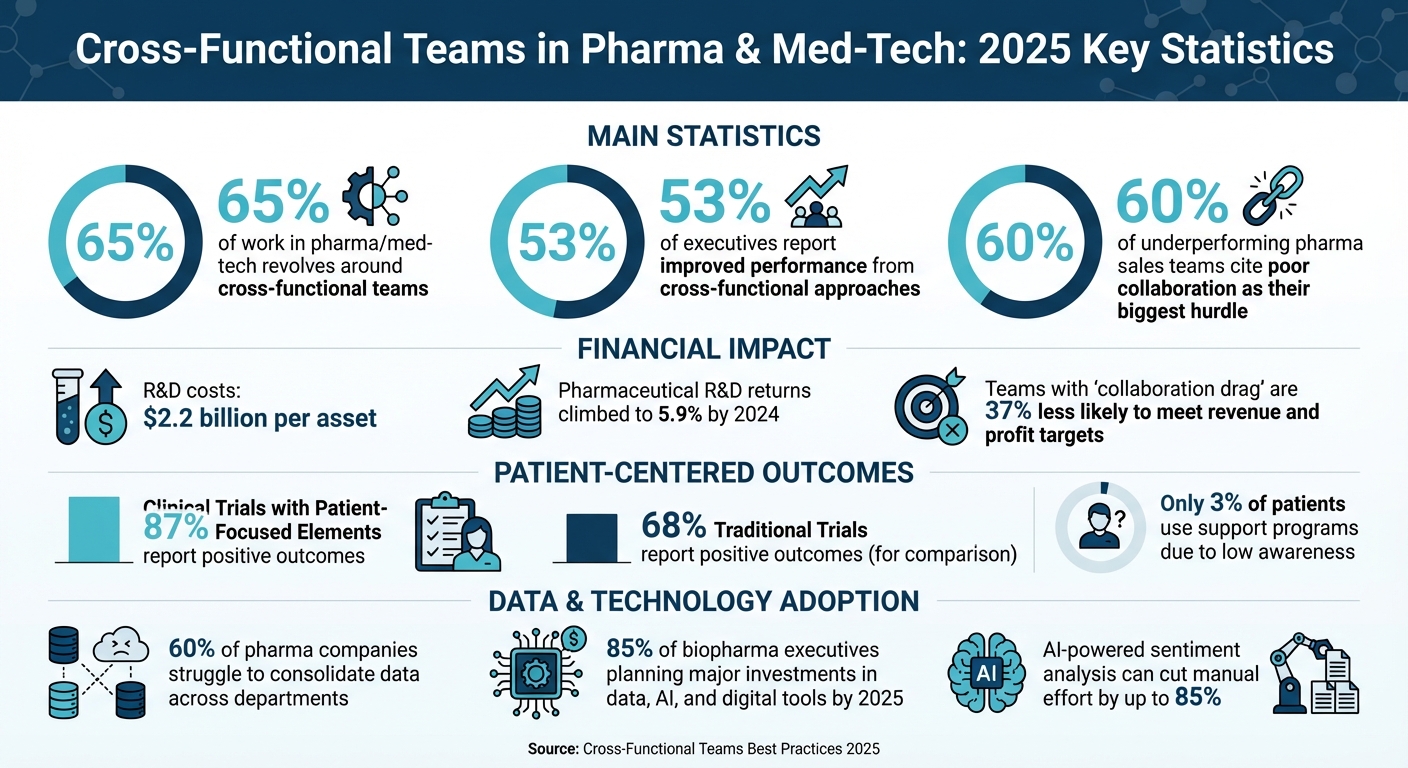
- 65% of work in these industries now revolves around cross-functional teams.
- 53% of executives report improved performance from this approach.
- Shared goals and clear missions are critical - misaligned teams face costly setbacks.
- Tools like RACI matrices, phase-gate reviews, and live traceability matrices help balance speed with compliance.
- Patient insights are key: Only 3% of patients use support programs due to low awareness.
To succeed, companies must focus on clear goals, structured collaboration, and patient-centered strategies. By aligning diverse teams and leveraging tools effectively, organizations can drive better outcomes and stay competitive.
Cross-Functional Teams in Pharma: Key Statistics and Impact 2025
Leading Cross-Functional Pharma Teams with Ian Wilcox & Megan Marshall - A Podcast About Leadership
sbb-itb-8f61039
Set a Clear Mission and Shared Goals
When cross-functional teams lack a unified mission, it’s no surprise that 60% of underperforming pharma sales teams cite poor collaboration as their biggest hurdle. The solution? Start by defining a clear, shared purpose that goes beyond individual departments. This approach helps break down the silos we discussed earlier. Instead of focusing on isolated metrics, successful teams ask: How does this benefit the patient?
With R&D costs exceeding $2.2 billion per asset, misaligned goals can lead to expensive setbacks. A shared language can prevent this. For instance, replacing department-specific jargon with metrics like "time from target identification to first patient dosing" helps streamline efforts, reduce duplication, and align priorities.
"Good science without governance is an expensive hobby. Leaders fund decisions, not personalities."
- Mukhtar Ahmed, Ph.D., Director of Project & Portfolio Strategy
The advantages of shared goals are already evident. By 2024, pharmaceutical R&D returns had climbed back to 5.9%. Companies are shifting away from narrow, department-focused bonuses toward team-based rewards tied to cross-functional achievements like regulatory submissions or launch readiness.
How to Create a Unified Vision
To build alignment, start by involving key departments - sales, finance, marketing, and compliance - early in the process. This ensures goals reflect both market realities and regulatory requirements.
One effective approach is using shared KPIs that foster accountability across teams. For example, tying customer satisfaction metrics to sales targets compels medical affairs, marketing, and sales to work together. Another strategy is adopting a "North Star metric", such as launch readiness milestones or adherence to time-to-IND goals, to connect daily tasks with broader business objectives.
"The final customer for all functions should be the patient."
- Michaela Pantaleoni, Chief Market Access Officer, Napo Therapeutics
Practical steps include implementing a Product Trio model. This team consists of a Product Manager (focused on the "why"), an Architect (focused on the "how"), and a Designer (focused on the "who/how it feels"). Together, they ensure both business value and technical feasibility. Another tactic is milestone-based funding, which allocates resources based on progress at key checkpoints - like assay qualification or pilot CMC batches. This approach reduces sunk-cost bias and encourages disciplined decision-making.
Additionally, involving regulatory experts early - such as during Phase 3 trials - and equipping marketing teams with clear compliance checklists can significantly lower the chances of asset rejection.
These strategies have already delivered measurable success for leading pharmaceutical companies.
Examples of Mission-Driven Success in Pharma
Pharma companies with clear, unified missions have achieved impressive results.
In May 2025, Takeda launched the "INDIE" initiative under Senior Director Clemens Muller. This program brought together global and local medical, commercial, and market access teams to address specific challenges collaboratively.
Similarly, Napo Therapeutics, led by Chief Market Access Officer Michaela Pantaleoni, introduced a rare disease initiative that emphasized patient-centricity. Daily 15-minute standups and biweekly strategic updates ensured the team stayed aligned and agile.
Between 2024 and 2025, AstraZeneca used a disciplined framework - centered on "right target, right tissue, right safety" - to enhance R&D productivity. By sticking to this mission, the company made tough but necessary decisions about which projects to advance or halt, a vital capability given the enormous costs of each program.
These examples highlight a key takeaway: when every function aligns around clear, measurable goals, collaboration evolves from a buzzword into a genuine driver of success.
Structure Teams for Speed and Efficiency
Creating a clear team structure is key to speeding up decisions and ensuring compliance in the heavily regulated pharma and med-tech industries. The challenge? Building teams that can move fast while following strict regulations like the FDA's Quality Management System Regulation (QMSR), which takes effect on February 2, 2026. The way teams are structured directly impacts how governance and operational agility are achieved.
The first step is selecting an effective governance model. Large companies often rely on matrix structures, where employees report to multiple managers across different functions. This setup encourages innovation but requires well-defined roles to avoid confusion. On the other hand, smaller companies and startups thrive with flat, non-hierarchical models. For example, at Napo Therapeutics in May 2025, Chief Market Access Officer Michaela Pantaleoni spearheaded a rare disease initiative using daily 15-minute standups and bi-weekly leadership updates. This lean, streamlined approach enabled quick pivots without getting stuck in red tape.
Another effective approach is the Product Trio model, which unites a Product Manager (focused on the "why"), an Architect (focused on the "how"), and a Designer (focused on the "who"). This model ensures that business goals, technical feasibility, and user experience are all addressed from the outset. For regulated industries, tools like RACI matrices (Responsible, Accountable, Consulted, Informed) and phase-gate reviews help ensure cross-functional alignment and proper sign-offs at key milestones.
Governance Models for Small vs. Large Organizations
The size of an organization plays a big role in determining the best governance practices. Here's a comparison:
| Feature | Small Organizations / Startups | Large Pharmaceutical Enterprises |
|---|---|---|
| Structure | Non-hierarchical; Agile "Product Trio" | Matrix structure; Formal phase-gate reviews |
| Decision-Making | Fast, often through daily standups | Structured milestone reviews with cross-functional input |
| Key Challenges | Limited resources; Building foundational quality systems | Functional silos; Conflicting priorities; Managing data overload |
| Compliance Focus | Establishing initial Design History Files (DHF) | Harmonizing global standards (GDPR, HIPAA, QMSR) |
| Outcome | High speed and adaptability; "fail fast" capability | Scalable, repeatable processes; High audit readiness |
These governance practices promote collaboration across functions, ensuring all teams work together toward patient-focused goals. For instance, Takeda's "INDIE" initiative in May 2025 used structured funnels to filter insights from medical, commercial, and market access teams, separating actionable intelligence from excess data.
Once governance models are in place, the next challenge is balancing flexibility with regulatory demands.
Balance Flexibility with Compliance Requirements
Navigating regulations like HIPAA, GDPR, and FDA guidelines while remaining agile requires careful planning. Compliance shouldn't just be a final step - it needs to be integrated from the beginning. Cross-functional collaboration during Phase 3 trials is one way to identify regulatory hurdles early, ensuring timelines for messaging stay on track.
A practical tool for this is a live traceability matrix, which connects design inputs (user needs) to outputs (specifications) and verification activities (test results). This tool keeps teams audit-ready while allowing the product to evolve. In med-tech, the Design History File (DHF) becomes the central hub for tracking all development decisions, ensuring transparency and defensibility during FDA inspections.
"Risk management under ISO 14971 is not a standalone exercise; it is a cross-functional discipline that drives design choices."
- Denis Katz, MD, Salience Clinical, LLC
Digital tools like Veeva Vault streamline version control, cutting down on email back-and-forth and creating a reliable audit trail. Additionally, providing marketing teams with regulatory checklists early in the creative process can significantly reduce the chances of asset rejection.
The takeaway? Teams can achieve both speed and compliance. By building the right structure, using shared tools, and integrating compliance from the start, cross-functional teams can innovate while maintaining regulatory confidence.
Improve Communication Across Functions
Clear communication between R&D, marketing, and patient engagement teams is essential for effective collaboration in the pharma and med-tech industries. The problem? These teams often work in silos, using different tools, metrics, and even terminology. When communication breaks down, patients ultimately feel the impact.
The starting point for fixing this is creating a standardized way to share information. Kimberley Chiang, Vice President of Biopharma Commercial Solutions at CoverMyMeds, explains:
"Within pharma, we don't have standardized data protocols. That makes it difficult to talk the same language amongst our groups".
Using shared metrics - like Customer Lifetime Value (CLV) and Net Revenue Retention (NRR) - can help align everyone around common goals.
Beyond standardization, early collaboration between teams is critical. Getting patient engagement and commercial teams involved from the start ensures that treatments are developed with both patient needs and market realities in mind. Instead of R&D working in isolation and then handing off a product to marketing, integrating teams early avoids mismatches between technical solutions and patient expectations. Rafael Oliveira, Group Product Manager at PartnerStack, highlights this point:
"Great companies set goals for the whole company, not for specific teams. It's important that business leadership communicates priorities and how each area can work together toward those goals".
Teams that struggle to collaborate - experiencing what’s called "collaboration drag" - are 37% less likely to meet revenue and profit targets. On the flip side, organizations that use tools like Service Level Agreements (SLAs) and RACI matrices to clarify decision-making and handoffs often see faster execution and fewer issues during transitions.
Digital Tools That Support Team Collaboration
Digital platforms can dramatically improve how cross-functional teams work together. Replacing scattered emails and documents with centralized tools creates a "single source of truth", keeping everyone on the same page. Platforms like Jira and Trello allow real-time task tracking, so R&D knows when marketing needs final specs, and patient engagement teams stay informed about new developments. Communication tools like Slack and Microsoft Teams make it easy to connect instantly, even across different locations [14, 15].
Tifenn Dano-Kwan, Chief Marketing Officer at Amplitude, emphasizes the value of centralized systems:
"Typically, when you have a centralized platform where all teams look at the same trusted, clean data, that data set unifies and equalizes decisions, conversations, and opinions".
Analytics tools like Amplitude and Gong can further enhance collaboration by providing direct insights into patient challenges. These insights are accessible across teams, helping everyone stay aligned without needing endless meetings. For patient-focused alignment, platforms like PatientPartner connect prospective patients with mentors, offering real-time feedback that can shape both R&D and marketing strategies.
Security and compliance are non-negotiable. Modern tools increasingly use blockchain to ensure data integrity throughout the development process. However, adoption can be slow due to knowledge gaps about interoperability solutions among providers and pharmacists. Starting small with a single, visible project can demonstrate the value of these tools before scaling up.
While digital tools are powerful, they’re not a replacement for human interaction. They work best when paired with regular check-ins to maintain alignment.
Schedule Regular Cross-Functional Check-Ins
Frequent, structured check-ins are essential for keeping teams aligned and addressing challenges early. The key is finding the right rhythm and purpose for these meetings. A tiered approach often works well: weekly one-on-ones between technical and design leads, monthly syncs with sales and patient engagement teams to gather customer insights, and quarterly reviews with marketing to plan strategic launches.
These meetings should focus on decision-making, not just updates. Rafael Oliveira from PartnerStack stresses the importance of using feedback effectively:
"If I'm sharing feedback and don't know if it's being used, I get discouraged to share more".
Effective check-ins give teams the chance to explain priorities and trade-offs, helping marketing and sales set realistic expectations.
Psychological safety is the foundation of productive check-ins. A 2024 BCG study found it to be the top driver of team success. When team members feel safe admitting mistakes or asking questions, problems get resolved faster. Leaders can model this by openly discussing risks and trade-offs during meetings.
Short, focused stand-ups can address immediate blockers, while monthly "Voice of Customer" sessions - where patient engagement and sales teams share direct patient feedback with R&D - can make abstract data feel more real [13, 11]. Establishing clear handoff protocols through SLAs, which define response times and quality standards, ensures smoother transitions and minimizes friction throughout the patient journey.
Use Patient Insights to Guide Team Decisions
Pharma and med-tech teams that truly embrace patient-centered approaches go beyond just lip service - they actively incorporate patient voices at every stage of decision-making. The shift from seeing patients as passive recipients to engaging them as active partners has reshaped collaboration. Instead of simply designing treatments for patients, leading organizations now work with them, leveraging their insights to drive research and innovation. This change isn't just theoretical; it's delivering results. For example, 87% of clinical trials with patient-focused elements report positive outcomes, compared to 68% for traditional trials.
However, integrating patient insights into decision-making isn't always straightforward. One of the biggest hurdles is the siloed nature of data within pharmaceutical organizations. Around 60% of companies struggle to consolidate data across departments like medical affairs, commercial teams, and market access. When teams rely on separate data sets, understanding patient needs holistically becomes a challenge. A centralized repository for patient insights can bridge this gap, ensuring everyone works from the same playbook.
Integrate Real-Time Patient Feedback
Real-time feedback is a game changer for turning raw data into actionable steps. Thanks to digital platforms, patients - especially those with mobility challenges or chronic conditions - can now share their input asynchronously, at their convenience. This ensures all voices, including those from underrepresented groups, are heard equally. Tools like PatientPartner (https://patientpartner.com) connect patients with AI mentors who share firsthand experiences, allowing cross-functional teams to gain immediate insights into patient concerns, challenges with adherence, and treatment experiences.
AI-powered sentiment analysis takes this a step further. By processing open-text feedback from emails, SMS, and in-app prompts, it identifies urgent themes and separates valuable insights from background noise. This automation can cut manual effort by up to 85%, giving teams more time to focus on meaningful action.
Takeda’s "INDIE" initiative offers a practical example. This program collects insights from various sources, prioritizes them by asking “so what?” for each data point, and distributes actionable intelligence to the right teams. Clemens Muller, Senior Director at Takeda, highlights the importance of connecting insights across all levels:
"It's essential to define what challenge we're solving and ensure that insights from medical, commercial, and market access are connected globally, regionally, and locally".
But gathering feedback is only half the battle. Closing the loop is just as critical. Lance Hill, CEO of Within3, cautions:
"If insights are just dropped into a meeting with no follow-up, it becomes a chore instead of a strategic process".
Teams must track how specific patient insights shape decisions and communicate those outcomes back to the stakeholders who provided the original feedback. This not only strengthens trust but also ensures that patient input leads to tangible improvements.
Apply Data-Driven Approaches for Patient Outcomes
Data analytics uncovers patterns that might otherwise go unnoticed. Take Janssen, for example. In October 2025, the company collaborated with patients to co-design clinical trial schedules for Spravato, a treatment for depression. By incorporating hybrid in-person and virtual visits based on patient preferences, they reduced participant burden and improved retention rates.
AstraZeneca took a similar approach with its asthma drug, Fasenra. By partnering with patient advocacy groups to create recruitment materials and host pre-trial webinars, they sped up recruitment and improved adherence. Meanwhile, Takeda used ethnographic research to map the patient journey for Entyvio, its ulcerative colitis treatment. This helped them address specific pain points, boosting participant satisfaction and reducing trial dropout rates.
Despite these successes, there's still work to be done. Only 3% of patients currently use available support programs, largely due to a lack of awareness. Data analytics can pinpoint where patients disengage in their journey and identify barriers like complex enrollment processes or poorly timed communications. Armed with this knowledge, teams can tailor solutions - whether that’s simplifying sign-ups, adjusting messaging, or offering additional services like education and logistical support.
AI-driven tools add another layer of insight. By analyzing scientific literature, social media, and claims data, these tools can reveal unexpected trends. For instance, in some therapeutic areas, mental health professionals - not the anticipated specialists - are leading treatment discussions. Insights like these help research, marketing, and patient engagement teams align their strategies with actual treatment patterns.
With 85% of biopharma executives planning major investments in data, AI, and digital tools by 2025, the infrastructure for data-driven decision-making is becoming the norm. The organizations that thrive will be those that don’t just collect patient data but also turn it into coordinated, actionable strategies across all teams - reinforcing the collaborative and agile structures needed for success.
Key Takeaways for 2025 and Beyond
Leading cross-functional teams in pharma and med-tech relies on four interconnected practices. At the core is mission alignment - when all departments focus on shared KPIs, such as "Time-to-IND" or "Patient Support Program Utilization", the emphasis shifts from individual wins to achieving better patient outcomes. Rafael Oliveira, Group Product Manager at PartnerStack, captures it well:
"The key component of cross‐collaboration is that it's not product‐goal, partnership‐goal and sales‐goal. There's a company goal above all and everyone is part of it".
This shared purpose creates the foundation for agile operational structures. Traditional hierarchies are giving way to smaller, nimble "pods" of 8–12 people dedicated to specific programs. These pods have played a key role in improving R&D returns to 5.9% in 2024 by reducing coordination delays and clarifying responsibilities. While pods encourage quick action, milestone funding and strict review gates ensure regulatory compliance remains intact.
Effective communication is another critical pillar. It’s more than just scheduling meetings - it’s about fostering continuous alignment. Daily 15-minute standups and digital tools like Veeva Vault streamline approvals and help surface challenges early. This reduces the risk of miscommunication and ensures insights flow seamlessly across teams.
Rounding out the system is a strong emphasis on patient insights. Decisions must be rooted in patient feedback, and creating a centralized repository for this data ensures it’s accessible across all functions. Tools like PatientPartner (https://patientpartner.com) offer real-time mentorship insights, bridging the gap between patient experiences and organizational priorities. Michaela Pantaleoni, Chief Market Access Officer at Napo Therapeutics, emphasizes this point:
"The final customer for all functions should be the patient".
Organizations that integrate mission alignment, agile structures, effective communication, and patient insights into their processes will be better positioned to deliver meaningful outcomes for patients.
FAQs
What’s the best way to pick shared KPIs for a cross-functional team?
The most effective way to choose shared KPIs for a cross-functional team is by tying them directly to the company’s broader goals. Make sure priorities are well-communicated and encourage collaboration between departments. This creates a shared focus, making it easier for team members to align their efforts and achieve collective objectives.
How can teams move fast without risking FDA, HIPAA, or GDPR compliance?
Teams can operate efficiently while staying compliant by implementing clear decision-making frameworks, promoting transparency, and agreeing on shared goals that focus on patient safety and data security. Encouraging collaboration and accountability across departments helps maintain a balance between speed and adherence to regulatory standards.
How do we turn patient feedback into actions the whole team can track?
To make patient feedback meaningful for your team, start with a structured collection process. Automated, multi-channel methods can help gather responses efficiently. Then, leverage AI tools to sift through the data, pinpoint recurring themes, and rank issues based on priority. Share these insights on a centralized platform so everyone on the team can access them easily.
Encourage collaboration by fostering discussions across departments, ensuring feedback is integrated into your planning process. Finally, establish clear metrics to measure progress. This keeps the team accountable and ensures ongoing improvements that align with what patients truly need.




